Repair vs Regeneration: Understanding the Critical Differences in Tissue Healing
When our bodies experience injury or damage, they initiate fascinating biological processes to heal and restore function. Two primary healing mechanisms come into play: repair and regeneration. Though often mentioned together, these processes are distinctly different in their approach, capabilities, and outcomes for tissue healing. Have you ever wondered why some injuries heal without a trace while others leave permanent scars?
Understanding the difference between repair and regeneration isn't just academic curiosity—it's crucial knowledge that underpins modern medical treatments and the emerging field of regenerative medicine. The human body's healing capabilities vary dramatically depending on the tissue type and extent of damage, determining whether complete restoration is possible or if permanent changes will remain.
In this comprehensive guide, we'll explore how tissue healing mechanisms work, what distinguishes repair from regeneration, and why certain tissues can regenerate while others cannot. Let's dive into the fascinating world of tissue healing to understand what happens beneath our skin when injury strikes.
What Is Tissue Repair?
Tissue repair is a fundamental healing process that restores both tissue architecture and function after an injury occurs. It represents the body's comprehensive response to damage and works through two distinct mechanisms: regeneration and replacement. When damage occurs, repair processes immediately begin working to restore normal structure and function.
The repair process is like your body's emergency response team, rushing to the scene when injury strikes. Initially, inflammation signals the damage, bringing immune cells to clear debris and fight potential infections. Blood clotting creates a temporary scaffold, and then the real reconstruction work begins through cell proliferation and matrix deposition.
Importantly, repair involves both regeneration (the regrowth of identical tissue) and replacement (the substitution of damaged tissue with fibrous connective tissue). The balance between these two processes determines whether the healed tissue will function normally or experience some permanent changes. In many cases, especially with severe injuries, repair results in scar formation rather than perfect restoration of the original tissue.
The tissue repair process involves multiple phases including inflammation, proliferation, and remodeling.
For example, when a heart attack (myocardial infarction) damages cardiac tissue, the body repairs the injury primarily through replacement. Since heart muscle cells (cardiomyocytes) have very limited regenerative capacity, the damaged area is replaced with a scar composed of connective tissue. While this repair preserves the overall heart structure, it can impair cardiac function since the scar tissue cannot contract like normal heart muscle.
Repair processes are remarkably versatile but have limitations. They're highly effective at restoring epithelial layers like skin but may struggle with complex organs and specialized tissues. The outcome of repair depends on numerous factors including the type of tissue damaged, the extent of injury, the individual's age, overall health, and genetic factors that influence healing capabilities.
What Is Tissue Regeneration?
Regeneration represents the ideal healing outcome—a complete restoration of damaged tissue to its original state with normal structure and function. Unlike repair with scarring, true regeneration results in tissue that's indistinguishable from the pre-injured state. This remarkable process occurs primarily through the proliferation of cells within the damaged tissue, effectively replacing what was lost with identical new tissue.
The regenerative capacity of tissues varies dramatically throughout the body. Some tissues demonstrate extraordinary regenerative abilities, while others have virtually none. This variation largely depends on the tissue's cellular proliferation capabilities and the presence of stem cell populations that can replenish damaged cells.
Hematopoiesis is an example of continuous regeneration, producing billions of new blood cells daily from stem cells.
Tissues with impressive regenerative capacity fall into the category of "continuously-dividing" or "labile" tissues. These include:
- Skin epithelium, which can rapidly heal superficial wounds
- Gastrointestinal epithelium, replacing itself approximately every 5-7 days
- Hematopoietic tissue in bone marrow, generating billions of new blood cells daily
- Liver tissue, capable of regenerating up to 70% of its mass after damage
These highly regenerative tissues contain pools of stem cells that remain self-renewable and proliferative throughout life. When injury occurs, these stem cells activate, divide, and differentiate to replace the damaged cells with functionally identical new ones.
Some tissues exhibit intermediate regenerative capacity and are classified as "quiescent" or "stable" tissues. These include kidney parenchymal cells, pancreatic cells, fibroblasts, smooth muscle cells, endothelial cells, and lymphocytes. While not continuously dividing, these cells can reenter the cell cycle upon stimulation from injury, allowing for some degree of regeneration when needed.
Permanent Tissues: The Limitation of Regeneration
Not all tissues in the human body possess regenerative capabilities. Some specialized cells, known as "permanent tissues," have left the cell cycle permanently and cannot divide to replace damaged cells. These tissues include:
- Cardiac muscle (heart tissue)
- Skeletal muscle (to a significant degree)
- Neurons in the central nervous system (brain and spinal cord)
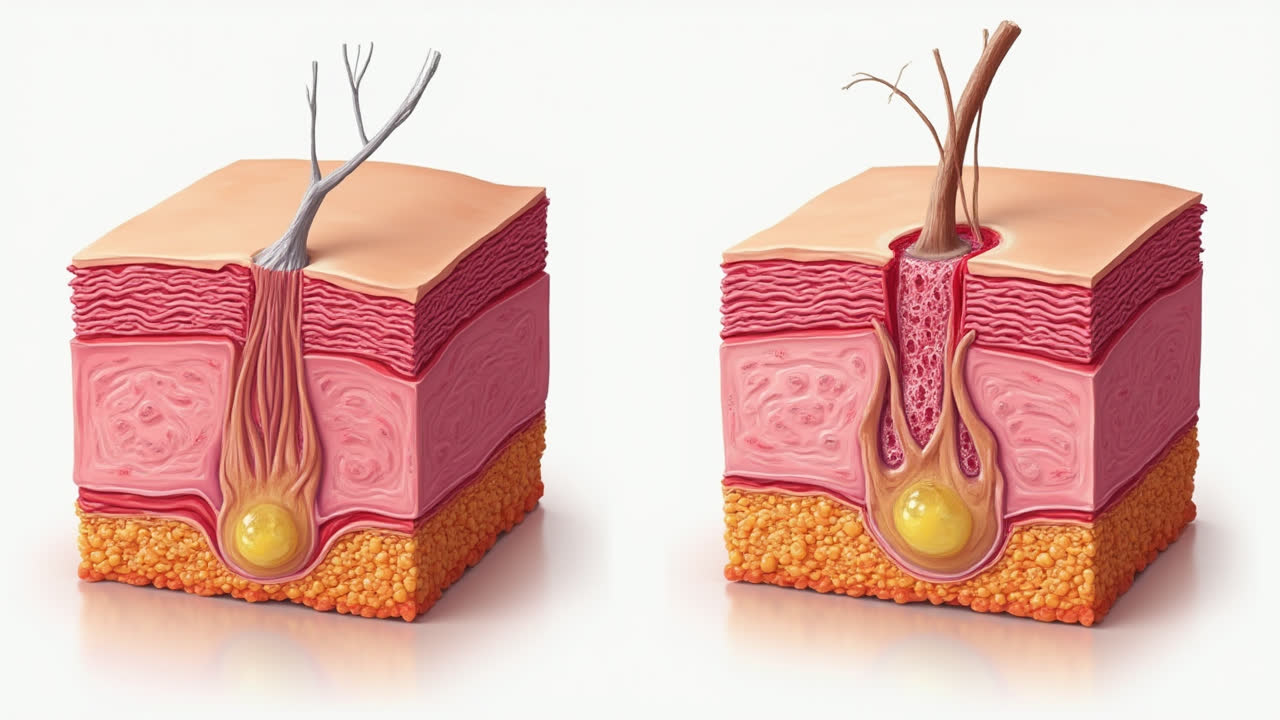
When these permanent tissues suffer injury, they cannot undergo true regeneration. Instead, repair occurs primarily through replacement—laying down connective tissue to fill the gaps left by damaged cells. This replacement process invariably leads to scarring and can result in permanent functional impairment.
For instance, when a stroke damages brain tissue, the lost neurons cannot regenerate. The body repairs the area by forming a glial scar (primarily composed of supporting cells called astrocytes), but this scar tissue cannot conduct electrical signals like neurons. Similarly, damage to heart muscle from a heart attack leaves a permanent scar that cannot contract like normal cardiac muscle, potentially compromising heart function.
This limitation of regeneration in permanent tissues explains why injuries to the brain, spinal cord, and heart often result in permanent disability. It also highlights why these areas are major focuses of regenerative medicine research—scientists are seeking ways to overcome these natural limitations and induce regeneration in tissues that normally cannot regenerate.
Key Similarities Between Repair and Regeneration
While repair and regeneration represent different healing outcomes, they share several important commonalities in their fundamental purpose and cellular mechanisms:
- Both processes are initiated in response to tissue injury
- They work toward the common goal of reestablishing tissue structure and function
- Both processes involve receptor-mediated signal transduction to coordinate cellular activities
- Various growth factors and signaling molecules mediate both repair and regeneration
- Inflammation plays a critical initiating role in both healing processes
- Both require vascular support to provide nutrients and oxygen to healing tissues
These shared characteristics reflect the evolutionary conservation of healing mechanisms across different tissues. Despite their differences in outcome, repair and regeneration utilize many of the same cellular tools and molecular pathways. Both processes require precise coordination of multiple cell types, signaling molecules, and extracellular matrix components to successfully restore tissue integrity.
Understanding these similarities has practical implications for medical treatments. Therapies that enhance these common healing pathways—like growth factor treatments, stem cell therapies, and scaffolds that support cell migration—can potentially improve both repair and regeneration outcomes across different tissue types.
Comparative Analysis: Repair vs. Regeneration
| Comparison Factor | Tissue Repair | Tissue Regeneration |
|---|---|---|
| Definition | Restoration of tissue architecture and function after injury through regeneration and replacement | Complete restoration of damaged tissue to its normal state through new growth |
| Primary Mechanism | Combination of regeneration and replacement (scarring) | Cell proliferation without scarring |
| Type of Damage Addressed | Major tissue damages, severe injuries | Minor tissue damages, superficial injuries |
| Scar Formation | Often involves formation of scar tissue | No scar tissue formation |
| Functional Outcome | May result in structural abnormalities that impair function | Restores original function with no permanent impairment |
| Tissue Types | Occurs in all tissue types, especially permanent tissues | Primary healing in continuously-dividing and stable tissues |
| Cell Source | Primarily fibroblasts (for replacement) and local cells | Stem cells and specialized proliferative cells |
| Time Required | Often slower, especially with scar remodeling | Generally faster in tissues with high proliferative capacity |
This comparison highlights the fundamental differences between these two healing processes. While regeneration represents the ideal outcome with complete restoration of tissue structure and function, repair through replacement (scarring) is often the reality for many injuries, especially in permanent tissues or cases of severe damage.
Modern medical interventions increasingly aim to shift the balance from repair with scarring toward regeneration when possible. This goal drives advances in regenerative medicine, tissue engineering, and therapeutic approaches that enhance the body's natural healing capabilities.
The Role of Stem Cells in Regeneration
Stem cells play a critical role in tissue regeneration, serving as the primary cellular source for replacing damaged or lost cells. These remarkable cells possess two defining characteristics: the ability to self-renew (make more stem cells) and the capacity to differentiate into specialized cell types. These properties make stem cells the foundation of regenerative capacity in tissues.
Different tissues contain various types of stem cells, each with specific regenerative capabilities:
- Totipotent stem cells (early embryonic cells) can form an entire organism
- Pluripotent stem cells can differentiate into cells from any of the three germ layers
- Multipotent stem cells can develop into multiple, but limited cell types
- Unipotent stem cells can produce only one cell type but retain self-renewal capacity
In adult tissues, resident stem cells typically exist in specialized microenvironments called niches that maintain stem cell populations in a quiescent state until activated by injury signals. Upon activation, these stem cells proliferate and differentiate to replace damaged cells, driving the regenerative process.
The abundance and activity of stem cells largely determine a tissue's regenerative potential. Tissues with robust regenerative capacity, like skin, intestinal epithelium, and bone marrow, contain substantial populations of active stem cells. In contrast, tissues with limited regeneration, like heart and brain, have few or inactive stem cells.
Understanding stem cell biology has revolutionized approaches to enhancing healing. Modern regenerative medicine strategies include transplanting stem cells, activating endogenous stem cells, creating artificial niches, and even reprogramming differentiated cells into stem-like states. These approaches aim to overcome natural regenerative limitations and promote healing in tissues that typically repair through scarring rather than regeneration.
Clinical Applications and Future Directions
The distinction between repair and regeneration has profound implications for clinical medicine. Traditional approaches to wound healing have often focused on supporting the repair process—keeping wounds clean, providing appropriate nutrition, and preventing complications. While these approaches remain valuable, modern medicine increasingly aims to promote true regeneration rather than repair with scarring.
Several cutting-edge approaches are being developed to enhance regenerative capacity:
- Tissue engineering combines cells, scaffolds, and growth factors to create functional tissue replacements
- Stem cell therapies introduce regenerative cells to damaged tissues to promote healing
- Growth factor treatments supply signaling molecules that stimulate cell proliferation and differentiation
- Gene therapy modifies genetic expression to enhance regenerative pathways
- Biomaterial scaffolds provide structural support for migrating cells during healing
These approaches have shown promising results in various applications, from treating chronic wounds and burns to addressing degenerative conditions affecting organs like the heart, liver, and nervous system. For instance, engineered skin substitutes now provide effective treatments for severe burns, while stem cell therapies show potential for regenerating damaged heart tissue after myocardial infarction.
The future of regenerative medicine looks toward increasingly sophisticated approaches that combine multiple strategies to overcome the natural limitations of healing. Research into cellular reprogramming, 3D bioprinting, and biomimetic materials continues to expand the possibilities for regeneration in tissues that typically heal through repair with scarring.
As our understanding of the molecular and cellular mechanisms governing repair and regeneration deepens, new therapeutic targets and approaches will emerge. The ultimate goal remains consistent: shifting the balance from repair with scarring toward complete regeneration, restoring both structure and function without permanent impairment.
Frequently Asked Questions About Repair and Regeneration
Why can some tissues regenerate while others cannot?
The regenerative capacity of tissues depends primarily on two factors: the presence of stem cells and the ability of specialized cells to reenter the cell cycle and proliferate. Continuously-dividing tissues like skin, intestinal lining, and bone marrow contain abundant stem cells that regularly replace cells, giving these tissues excellent regenerative potential. In contrast, permanent tissues like heart muscle and neurons in the central nervous system have cells that have permanently exited the cell cycle and contain few or no resident stem cells. Additionally, the local environment, including signaling molecules, immune responses, and extracellular matrix composition, influences whether regeneration or repair with scarring occurs. Evolutionary factors also play a role—mammals generally have more limited regenerative capacity than some amphibians and fish that can regrow entire limbs or organs.
Can medical treatments convert repair with scarring to regeneration?
Modern regenerative medicine approaches increasingly aim to convert repair with scarring to true regeneration, though complete success remains challenging. Several strategies show promise: stem cell therapies introduce regenerative cells to damaged tissues; growth factors stimulate cell proliferation and differentiation; biomaterials provide scaffolds that guide tissue formation; and gene therapies modify cellular expression to enhance regenerative pathways. For instance, platelet-rich plasma treatments deliver concentrated growth factors to accelerate healing in tendons and ligaments, while engineered skin substitutes containing living cells promote regeneration in severe burns. While these approaches can significantly improve healing outcomes and reduce scarring, complete conversion from repair to regeneration—especially in tissues like heart, brain, and spinal cord—remains an active area of research rather than clinical reality.
How does aging affect the balance between repair and regeneration?
Aging significantly shifts the balance from regeneration toward repair with scarring across most tissues. This shift occurs due to several age-related changes: stem cell populations decline in number and function; cellular proliferation capacity decreases; growth factor production and responsiveness diminish; chronic low-grade inflammation increases; and extracellular matrix composition changes, becoming less supportive of regeneration. Practically, this means older individuals experience slower healing, increased scarring, and less complete functional recovery after injuries. For example, a skin wound that might heal with minimal scarring in a child often results in more prominent scarring in an elderly person. Similarly, muscle injuries in younger people recover more completely than in older individuals. Understanding these age-related changes has spurred research into therapies that might rejuvenate aging tissues' regenerative capacity, including approaches targeting senescent cells, enhancing stem cell function, and modifying the inflammatory response.
Conclusion
The distinction between repair and regeneration represents one of the most fundamental concepts in tissue healing. While both processes aim to restore tissue after injury, they follow different paths and achieve different outcomes. Regeneration completely restores tissue structure and function through cell proliferation without scarring, while repair combines regeneration with replacement (scarring) to restore basic integrity, often with some functional compromise.
The body's ability to regenerate varies dramatically across different tissues, with continuously-dividing tissues showing robust regenerative capacity and permanent tissues relying primarily on repair with scarring. This variation explains why some injuries heal without a trace while others leave permanent damage.
Understanding the molecular and cellular mechanisms governing repair and regeneration continues to drive advances in regenerative medicine. By developing approaches that enhance the body's natural regenerative capacity or provide artificial means of regeneration, modern medicine increasingly aims to shift healing outcomes from repair with scarring toward complete regeneration.
As research progresses, the traditional boundaries between repair and regeneration may blur, with new therapeutic approaches enabling regeneration in tissues that naturally heal through repair. This exciting frontier promises improved outcomes for patients with injuries, chronic wounds, and degenerative conditions, bringing us closer to the ideal of perfect healing across all tissue types.